Atomic Model of Thomson
Thomson Atomic Model Theory and experiments With Its Characteristics
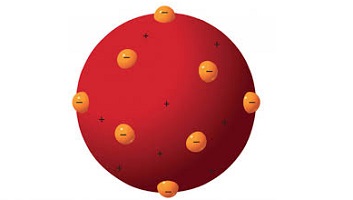
Thomson Atomic Model Theory and experiments: The atomic model of Thomson is the first theoretical description of the internal structure of atoms , proposed around 1900 by William Thomson (Lord Kelvin), strongly supported by Sir Joseph John Thomson, who had discovered (1897) the electron part negatively charged of each atom.

After Joseph John Thomson discovered the electron, in 1897, it was determined that matter was composed of two parts: a negative and a positive. The negative part was made up of electrons, which were, according to this model, immersed in a mass of positive charge, such as raisins in a cake or grapes in jelly.
Subsequently, Jean Perrin proposed a modified model based on Thompson’s model, where “raisins” (electrons) were located on the outside of the “cake” (the positive charge).
What is Thomson Atomic Model Theory and experiments
Thomson argued that atoms are uniform spheres of positively charged matter in which electrons are embedded. Popularly known as the raisin pudding model, it had to be abandoned (1911) for both theoretical and experimental reasons in favor of the Rutherford Atomic Model, in which electrons describe orbits around a small positive nucleus.
Characteristics of the atomic model of thomson or postulates of the atomic model of thomson
- The atom has a neutral charge.
- There is a source of positive charge that neutralizes the negative charge of electrons.
- This positive charge is evenly distributed in the atom.
- In Thomson’s words: “negatively electrified corpuscles,” that is, electrons, are contained within the uniform mass of positive charge.
- Electrons could drift freely inside the atom.
- The electrons had stable orbits, an argument based on Gaussian Law. If the electrons moved through the positive “mass”, the internal forces within the electrons were balanced with the positive charge that was automatically generated around the orbit.
- The atomic model of jj thomson was popularly known in England as a plum pudding model, since the electron distribution proposed by Thomson was similar to the arrangement of plums in that dessert.
Details of Thomson Atomic Model Theory and experiments
To explain the formation of ions, positive and negative, and the presence of electrons within the atomic structure, Thomson devised an atom resembling a fruit cake. A positive cloud that contained the small negative particles (electrons) suspended in it.
The number of negative charges was adequate to neutralize the positive charge. In the event that the atom lost an electron, the structure would be positive; and if he won, the final charge would be negative.
In this way, he explained the formation of ions; but left unexplained the existence of electromagnetic radiation, such as x-rays and other light phenomena.
Thomson and the electron
At the end of the 19th century, scientists began to notice behaviors in the field that could not be explained by the atomic model proposed by Dalton, such as the one carried out by Millikan in the mid-19th century, which consisted of dropping a drop of oil through an electric field in a vacuum closed tube, which determined that the minimum charge that the drop of oil acquired was equal to 1.6 × 10 ^ -19 C, which along with other electromagnetic experiences They gave the idea that the “indivisible spherical” model proposed by Dalton was wrong and that there was a charged particle that could be affected by changes in the electric field.
After these discoveries, scientists began trying to give an explanation to these phenomena. The most important was that made by Thomson in the late nineteenth and early twentieth centuries, which consisted of the use of a tube called “cathode ray tube” (Image 2), which consisted of two electrodes attached to a source of high voltage, connected in a vacuum closed tube.
Bypassing electric current from the power source to the anode (negative electrode) it was seen that a luminous gas was directed from the anode to the cathode (positive electrode).
This ray was called a cathode ray and was the one studied by Thomson, which subjected this ray to an electromagnetic field, observing that the ray deviated from its straight line towards the positive end of the field, which added to the knowledge that Opposite charges are attracted, he concluded that the cathode ray was composed of negative particles from the anode, which he called electrons.
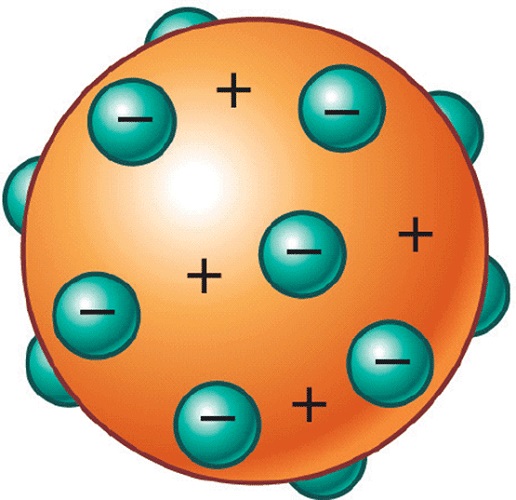
After this discovery, Thomson formulated a new atomic model that was called “raisin cake” (represented approximately in Image 3), which was that electrons were embedded in a positively charged sphere with the same charge intensity (this it was the first time that the electro-neutrality of the atom was proposed), in addition, Thomson could establish that the ratio between the charge of the electron (which was discovered by Millikan, explained above) and its mass was equal to 1,756 × 10 ^ 8.
JJ Thomson proposed the model that bears his name to explain the atomic structure. This consisted of a sphere of positively charged non-uniform matter, where the negative particles were inserted, that is, the electrons, hence this model is also known as “raisin pudding”, for the similarity with this sweet English.
The English physicist conducted a series of three experiments with cathode ray tubes, in his third test Thomson reached advanced conclusions, calling “corpuscles” to the particles that came from inside the electrode atoms, forming the cathode rays.
A cathode tube was a closed empty glass tube, to which the air was extracted and a gas was introduced at a reduced pressure. After this observation, he concluded that atoms are divisible.
Thanks to these experiments, he was also able to study the mass relationship between the particles that were attracted to the positive pole of the cathode tube.
He came to imagine that atoms were composed of these particles baptized as corpuscles within a lake full of positive charges, or what is the same, raisin pudding model.
This structure explained that matter was electrically neutral, since in atoms, according to Thomson, the positive charge was neutralized by the negative. These negative charges were sometimes evenly
distributed around the core, and in other cases the example of a positive charge cloud was used. Thanks to this discovery Thomson received the Nobel Prize in Physics in 1906.
Microscopically, this model can be said to have an open structure, since protons or positive charges are placed in the mass that defines the neutral charge of the atom.
This model was the first really atomic, although it was soon seen to be very limited.
Thomson’s model was discussed after the Rutherford experiment, when the nucleus was discovered, since this model cannot explain that the atom is formed by a dense nucleus and a part around it called the crust, so scientists such as Ernest Rutherford and Niels Bohr continued to investigate, and give theories about atoms.
Thomson experiments
Thomson Atomic Model Theory and experiments
Thomson’s first experiment

Description:
He investigated whether the negative charges of the cathode rays could be separated and used a means of magnetism.
For this experiment I build a cathode ray tube which at the end of the tube ends in two cylinders with grooves, the grooves were in turn connected to an electrometer.
With this method Thomson discovers that when the rays are magnetically deflected so that they cannot enter the grooves, the electrometer marks when registering little charge.
This led Thomson to conclude that the negative charge is inseparable from lightning.
Thomson’s second experiment

Description:
For this second experiment, JJ Thomson builds a cathode ray tube, achieving an almost perfect vacuum, at one of its ends it is coated with phosphorescent paint.
The intention of this experiment was to investigate whether these rays could be diverted with an electric field, it was known that in previous experiments this phenomenon had not been observed (this is very characteristic of charged particles).
With the creation of this tube in which at one end it was coated with phosphorescent paint, Thomson discovers that many rays could be bent with the influence of a magnetized field
Thomson’s third experiment

Description:
For the third experiment, Thomson founded the relationship between the mass of the cathode rays and the charge, for this he measures the amount that is deflected by a magnetic field and how much amount of energy charge is contained.
The mass / charge ratio that it finds is a thousand times higher than that contained in the Hydrogen ion, this indicates that the particles must be lighter or with much more charge.
Here Thomson takes a bold position: Thomson, to the cathode rays that were charged by particles he called “corpuscles” said corpuscles originated within the atoms of the electrodes, to what this meant, that the atoms must be divisible, imagine ” a sea ”totally full of positive charges in these corpuscles in the atom, that is why it is called and known by the name of raisin pudding to the Thomson model.
Continue with: